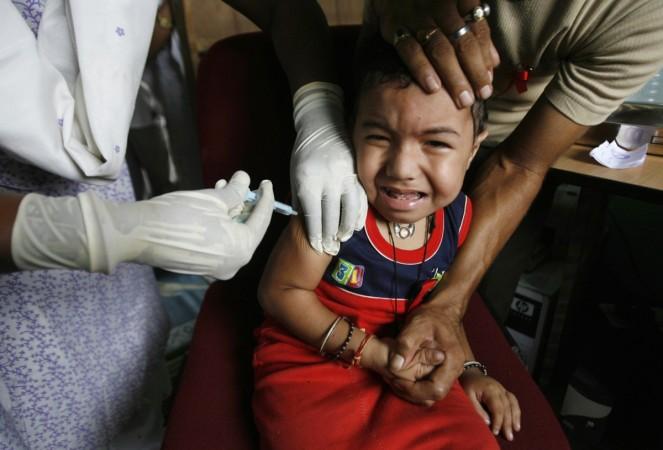
A Hyderabad-based biopharmaceutical company, Biological E. (BE) Limited, has received Rs 895 crore order from the union health ministry for a liquid pentavalent vaccine(LPV), ComBE Five, which would be inducted in the universal immunisation programme — Mission Indradhanush.
Mission Indradhanush, one of the flagship healthcare initiatives of the Narendra Modi government, aims to extend full immunisation coverage to at least 90 percent children of the country.
BE said in a statement it will supply the vaccine to the health ministry for a period of two years.
The company said the ComBE Five has received World Health Organisation's pre-qualification approval. The vaccine helps fight Diphtheria, Tetanus, Pertussis, Haemophilus Influenza Type b and Hepatitis B.
"More than 200 million doses of this 5-in-1 vaccine from BE have been administered to children around the world till date," the statement said.
It added the ComBE Five is a 5-in-1, convenient and safe Vaccine that provides effective protection for children (in the age group of six weeks to five years) against the five deadly diseases.
BE said it expected the vaccine to make a significant contribution in reducing the under five years mortality rate in India.
The company said it is investing Rs 500 crore for the expansion of its facilities over a period of three years to augment its production capacity of vaccines and complex generic medicines. This, according to BE, will generate 600 jobs.
"We are proud to be at the forefront of the Universal Immunization Programme of India since it aims at saving millions of precious lives of children. In fact, it signals the start of the much awaited countrywide rollout of LPV," Mahima Dutta, managing director of BE, said.
She added India is the most significant market and this contract would further contribute to BE's growth. "We are very proud to contribute to the Make in India initiative by becoming one of the world's largest vaccine manufacturers of LPV," she said.

















