Our immune system is programmed to fight invading pathogens and terminate them from our bodies. The idea of this very response promoting the growth of a pathogen is frightening, to say the least. Now, a new study has found that the attempts of the immune system to eliminate the deadly Salmonella bacteria from the gastrointestinal (GI) tract instead enables its colonization of the GI tract and shedding through feces.
According to researchers from the National Institutes of Allergy and Infectious Diseases (NIAID), Salmonella Typhimurium bacteria can use the defensive mechanism of intestinal epithelial cells (IECs) to replicate and grow. The study was published in the journal Cell Host & Microbe.
Delivering a 'Gut' Punch
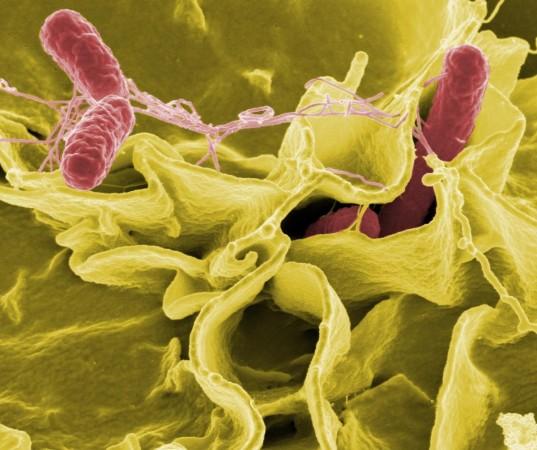
Salmonella Typhimurium (STm) is a pathogenic bacterium that lives in the intestines of humans and animals and is shed through feces. It causes gastroenteritis in human beings and other animals such as chickens, pigs, and cattle. The disease occurs mostly when contaminated food or water is consumed. Diarrhea, stomach cramps, and fever, are the most common symptoms of the infection and can resolve themselves without any particular treatment. In severe cases, antibiotics are used for the management of the infection.
Though the disease can be resolved clinically through medications, Salmonella can remain in the GI tract much longer after the abatement of the infection. When the bacteria are shed fecally, transmission to new hosts is facilitated. This transmission can be worse in the case of so-called 'super-shedders' who release large amounts of the bacteria through their feces.
According to the World Health Organization (WHO), Salmonella is one of the four key causes of diarrheal diseases globally. Approximately 550 million people fall sick each year due to the bacteria. One of the biggest challenges faced in the management of Salmonella is the emergence of antimicrobial resistance (AMR) in some of its serotypes.
Increase Rather Than Decrease?
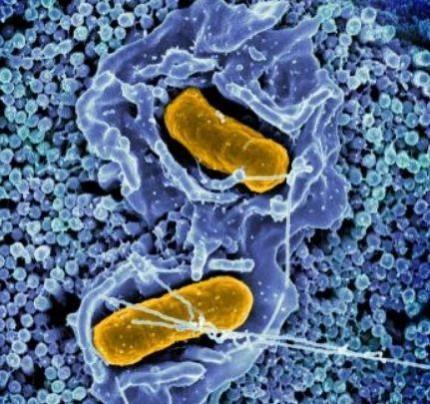
The focus of the authors was to understand how the Salmonella bacteria establishes itself and develops a strong footing in the mammalian GI tracts. A single layer of specialized cells known as intestinal epithelial cells, or IECs, forms the first line of defense within the GI tract. It serves as a physical barrier between the tissues beneath it and pathogens.
IECs absorb nutrients and serve as a crucial protective barrier that keeps pathogens away from deeper tissues that are more vulnerable. When these cells are invaded by bacteria, the cells are ejected into the hollow portion of the intestines known as the gut lumen.
In older studies, NIAID scientists had discovered that some Salmonella replicates quickly within the fluid portion of the intestinal epithelial cells called cytosol. Therefore, the team sought to ascertain whether the ejection of infected cells amplifies the growth of the bacteria (Cytosolic STm) rather than eliminating it.
Hijacking Protective Cells
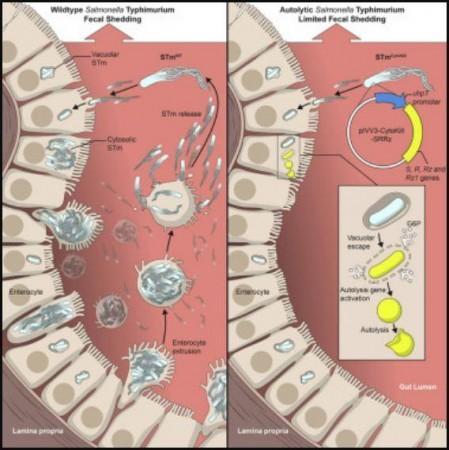
For the study, the authors developed genetically engineered Salmonella bacteria. This altered Salmonella (STm CytoKill) destructed when it interacted with the cytosol of epithelial cells but flourished normally in other environments such as the lumen of the intestine. Laboratory mice were infected with this self-destructing Salmonella variant next.
It was found that replication of the bacteria in the cytosol of the mice's intestinal cells is vital in the pathogen's colonization of the GI tract, and the intensification of fecal shedding. The researchers posit that through the hijacking of the epithelial cell response, Salmonella magnifies its capacity to invade and overwhelm the surrounding cells and prepare the intestine for shedding through feces.
According to the scientists, this serves as an example of the manner in which the burden of the host's immune response can lead to a pathogen's evolution, and vice versa. The findings of the study provide new insights into the development of medical interventions that can aid in softening the blow this pathogen can deliver.















