While COVID-19 was initially detected in human beings, the SARS-CoV-2 virus has been found to infect several animals. Over the past 20 months, the pathogen has been identified in various mammals such as big cats, dogs, and rodents, among others. On the other hand, researchers rely on animal models to learn more about the sickness and how it may potentially affect different animals. Bridging both these aspects, scientists have utilized an 'in vitro zoo' to study the effects of the coronavirus across.
Through a multi-institutional study, researchers developed a biobank of airway epithelial cell (AEC) culture models from 12 different animals. The models included both domesticated and wild animal cells. It was learnt that the SARS-CoV-2 virus replicated efficiently only in monkey and cat cell models, thereby, calling for vigilant surveillance of these species to avoid the creation of spillback reservoirs of the coronavirus.
"Our collection is unique, and thus far we are the first that have used such a large collection of advanced in vitro cell culture models from various domesticated and wildlife animal species to assess their susceptibility to SARS-CoV-2 infection," said Dr. Ronald Dijkman, corresponding author of the study, in a statement.
Building an 'In vitro Zoo'

In order to understand the effect of COVID-19, studies often utilize advanced in vitro cell culture models of the human respiratory tract. Therefore, the team aimed to create a biobank of similar models for various wild and domesticated animal species. They separated airway epithelial cells (AEC) from the tracheobronchial tissues in the lungs of several deceased animals. In total, cells from 12 varieties of animals were isolated— camel, cat, cattle, dog, ferret, goat, llama, pig, rabbit, rhesus macaque, and two species of neotropical bats.
This is of significance as the transmission of the virus has been witnessed in wild animals and species that humans have close interactions with such as pets. Therefore, these cells can be used to build well-differentiated AEC culture models that can be used to ascertain whether the SARS-CoV-2 virus can infect these animals.
Cats and Monkeys: Likely Spillback Reservoirs
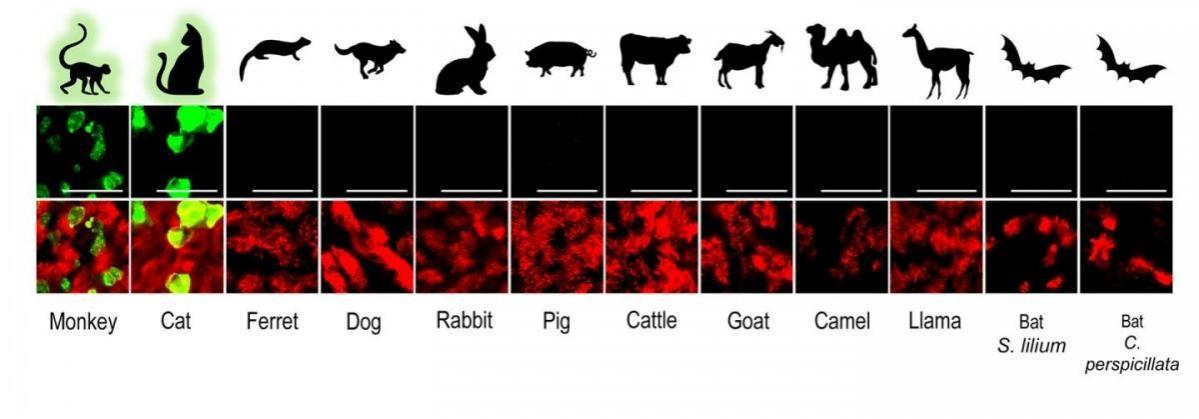
To investigate the susceptibility of different species to COVID-19, the scientists infected these 12 mammal AEC culture models with the SARS-CoV-2 virus and examined the progression. Employing whole viral genome sequencing, the authors found that the pathogen effectively multiplied only in the in vitro models of cats and rhesus macaques. It did so without having to adapt to these cells.
Hence, the findings indicate that certain species of cats and monkeys may be at a larger risk of contracting the viral infection. "Our findings, together with the reports from previously documented spillover events, indicate that close surveillance of these animals and other close relatives, whether they live in the wild, captivity or households, is necessary," explained Dr. Dijkman.
According to the authors, information from such models can prove beneficial to public health authorities in the surveillance of the virus at an animal-human interactional level. Particularly, it can enable the devising methods of streamlined early detection surveillance to observe animals that may potentially serve as spillback reservoirs for the novel coronavirus.
Dr. Dijkman stressed: "This will benefit the general public since it will help prevent new SARS- CoV-2 variants from developing in animal reservoirs and potentially being reintroduced into the human population, to which the current vaccines may not be protective."
Potential to Transform Animal Experimentation
The biggest advantage of the study's findings, however, is the potential to 'replace, reduce and refine' coronavirus research; particularly, animal experimentation. As the cells are acquired from animals post mortem and can be cultured in the lab to produce more, the need for experimenting on animals can be reduced or eliminated. The authors asserted that AEC culture models from various mammals can be employed to overcome the practical limitations of conventional in vivo experiments aimed at understanding interactions of the SARS-CoV-2 with different hosts.
"Our study shows that there is a lot of potential to replace, reduce and refine animal experimentation in the near future, and I hope that for basic fundamental research questions our results will convince researchers, pharmaceutical companies, and drug administration agencies to use advanced biologically relevant in vitro models prior to conducting animal experiments," expressed Dr. Dijkman.

















