Opioids are uncontrollably addictive drugs. The biggest danger associated with the abuse of these drugs is the high risk of overdose, which can often result in death. It is known that these fatalities are brought upon by disrupted breathing.
However, the exact mechanism through which these drugs suppress breathing is not known. Now, scientists have zeroed in on a set of neurons in the brainstem that play a crucial role in the repression of respiration during an overdose.
A multi-institutional study has reported the identification of specific receptors in a group of neurons, whose triggering results in opioid-induced respiratory depression (OIRD), the hampered breathing that causes death when an individual overdoses. The study found using an animal model that OIRD can be reversed by blocking these particular receptors.
"The underlying mechanism of why opiates slow down and depress the breathing rhythm has not been fully characterized. This knowledge can provide a stepping stone to better treatment options for OIRD," said Dr. Sung Han, senior investigator of the study, in a statement. The research was published in the journal Proceedings of the National Academy of Sciences.
Identifying Responsible Neurons
Drug abuse not only ruins one's health but also their quality of life. Along with this, the chances of these drugs causing their demise are significantly high. According to the WHO, around 0.5 million deaths are attributed to drug use annually. Over 70 percent of drug-caused deaths are associated with opioids, with over 30 percent of them resulting from overdoses.
Opioids such as morphine, heroin, and fentanyl, among others, work by binding with proteins known as opioid receptors present on neurons, eventually inhibiting their activity. As of now, naloxone is one of the only medications that is known to be able to stop the effects of opioids and counter an overdose.
Nevertheless, naloxone has its limitations. This includes a short duration of action—requiring multiple administrations—and blocking of opioid receptors across the body (including those that help control pain).
In order to devise improved approaches to tackle OIRD with more precision, the authors began looking for breathing neurons within the brain that also possessed opioid receptors. Through the current study, they discovered a group of neurons that express a particular variety of opioid receptors—the mu opioid or µ-opioid receptor.
These neurons are found in the breathing modulation center of the brainstem (the stalk-like structure that connects the cerebrum to the spinal cord). Following this, the team identified the role of the neurons in OIRD.
Countering OIRD and Improving Respiration
They used mice that were genetically modified to not contain µ-opioid receptors in the identified neurons. It was found that when these engineered mice were exposed to morphine, their breathing was not disrupted like it was in control mice.
Another important learning was that when these receptors were stimulated in control animals without the introduction of opioids, the mice still experienced symptoms of OIRD. Next, the researchers focused on finding ways through which OIRD could be reversed.
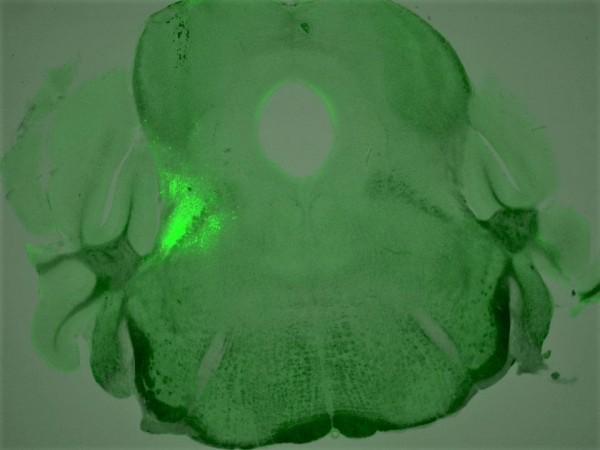
For this, they treated overdosed mice with compounds targeting other receptors present on the same neurons. The role of these receptors is opposite to that of the µ-opioid receptors. Therefore, they were activating those receptors rather than inhibiting them.
"We discovered four different chemical compounds that successfully activated these neurons and brought back the breathing rate during OIRD," noted Shijia Liu, first author of the study.
So what is next for the team? They intend to ascertain whether there is a role of other cell groups in OIRD. They will also study the link between the regulation of breathing and the perception of pain. This may potentially open new avenues in the development of targeted remedies for OIRD.
"We hope to explain the pain-breathing segregation at the molecular or microcircuit level. By doing that, we can try to restore breathing without touching analgesic effects of opioids," concluded Dr. Han.














