Cancer is the second leading cause of death worldwide, claiming over 9.6 million lives annually. While several treatments have helped patients fight off the disease, cancer has certain tools that it effectively uses to worsen their condition. Malignant tumors maximize their chances of survival and proliferation through the suppression of antitumor immune cells around them. Now, scientists have discovered a new process that can help counteract the immunosuppressive effects of tumors.
In a new multi-institutional study, researchers have found that certain factors secreted by lung cells called club cells possess anti-immunosuppressive properties. Using an animal model of lung cancer, the authors demonstrated that these club cell-secreted molecules inhibit potent immunosuppressive cells known as myeloid-derived suppressor cells (MDSCs). As a result, the number of antitumor T- cells (a type of lymphocyte) increased, and significantly enhanced the efficaciousness of immunotherapy.
"These club cell-secreted factors are able to nullify immune suppressor cells that otherwise help tumors escape an effective antitumor response. We're excited by the possibility of developing these club cell factors into a cancer treatment," said Dr. Vivek Mittal, co-senior author of the study. The research was published in the journal Nature Cancer.
Reversing Immune Suppression
Over the past few decades, cancer research has made strides in employing the body's immune system against the disease. Immunotherapy is one such example. Drugs known as immune checkpoint inhibitors (ICIs) can hinder or partially reverse the immunosuppressive effects of tumors. It has also been learned in recent years that ionizing radiation—a standard treatment for several forms of cancer—can further reverse this immunosuppression. This can improve the efficiency of ICI treatments.
Through the current study, the team aimed to ascertain the manner in which radiation gave rise to this immune-boosting action. In order to understand the underlying mechanism, a mice model of non-small-cell lung carcinoma—the most prevalent form of lung cancer—was utilized.
It was gleaned that at an average dose of radiation, the immune-enhancing effect peaked. This lead to a fourfold increase—to 40 percent—in the number of mice (treated with ICIs) who survived without tumors to the end of the observation period of two months.
Action of Club Cell Molecules
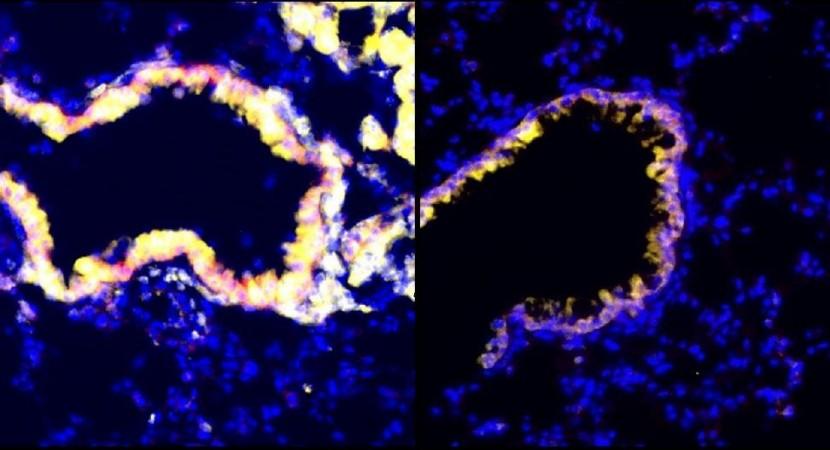
Next, the researchers found the answer to the important question—how does radiation result in this effect. It was discovered that radiation activates and stimulates the rapid increase of lung-resident Scgb1a1+ club cells. This particular group of cells is known to aid in the protection and repair of highly sensitive linings of the airway, partly by decreasing inflammation.
"It's possible that we see a peak stimulation of these cells at a particular radiation dose because a lower dose doesn't stress the cells enough, whereas a higher dose kills them," stated Dr. Nasser Altorki, co-senior author of the study.
Several molecules are secreted by the activated club cells. The authors learnt that radiation could be replaced by a "club cocktail" containing eight of these club cell-secreted molecules that can elicit the same ICI-enhancing outcome.
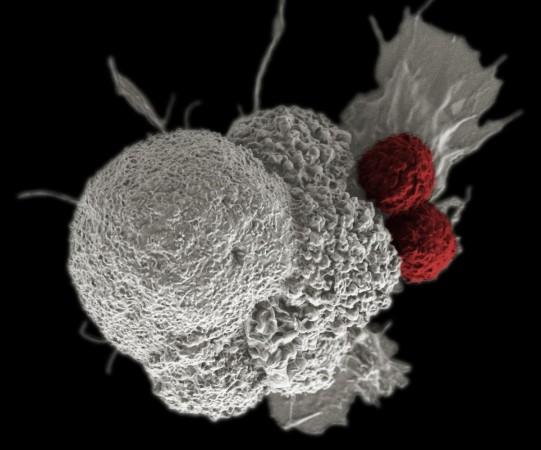
Adding to the promising list of learnings, the scientists also discerned that the inhibition of MDSCs by the club cell molecules is what gives rise to the immune-restoring effect. MDSCs are a known hurdle that impairs the efficiency of cancer immunotherapies. The inhibition of MDSCs caused the number of T-cells at the tumor site to increase and remarkably improved the effectuality of FDA-approved immunotherapy treatment.
Evidence of Clinical Translation
Armed with the new information, the team next sought to confirm whether these laboratory findings are relevant to human cancer. For this, they examined blood serum samples from lung cancer patients. These patients were part of a clinical trial conducted by Dr. Altorki and his colleagues. The trial evaluated the effectiveness of radiation plus ICI treatment and compared it to the administration of ICI treatment alone.
It was noted that the levels of CC10, a vital club cocktail molecule, were noticeably higher in most of the patients—5 out of 8—whose condition improved after the treatment. However, in patients whose condition did not improve (0 out of 9), no such elevation was observed. This suggested that CC10 can aid in the improvement of patients' outcomes.
Currently, the authors are working on ascertaining which of the molecules within the club cocktail are most essential in the suppression of MDSCs and amplifying the efficiency of cancer therapy. Additionally, they also intend to analyze whether MDSCs in other forms of tumors can be inhibited by these club cell molecules.
"We hope that these secreted molecules will be able to enhance treatments not just for non-small cell lung cancer patients but for patients with other cancers as well. These molecules may also be useful as biomarkers predicting the response to combined radiotherapy and immunotherapy," concluded Dr. Dingcheng Gao, co-senior author of the study.








